In this lab you will create a quantitative plot for lead fraction in a mixture of lead and sodium EDTA.
There are three links. One is for the lab instructions, a second is for an article on the lab and a third allowing a dry lab for the “calibration” curve from the spectra.
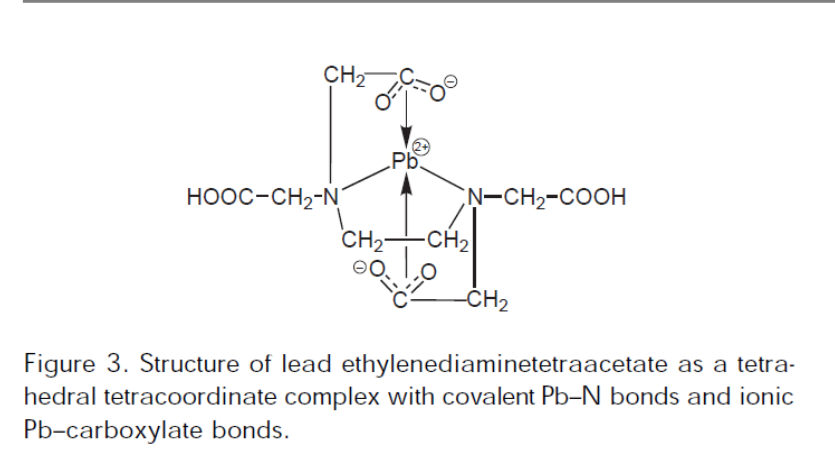
Students should choose a particular cm-1 to quantitate and be prepared to explain why. How will you record the peak? You should notice that there is a zero % mixture for comparison to which the magnitude of the peak is compared.
